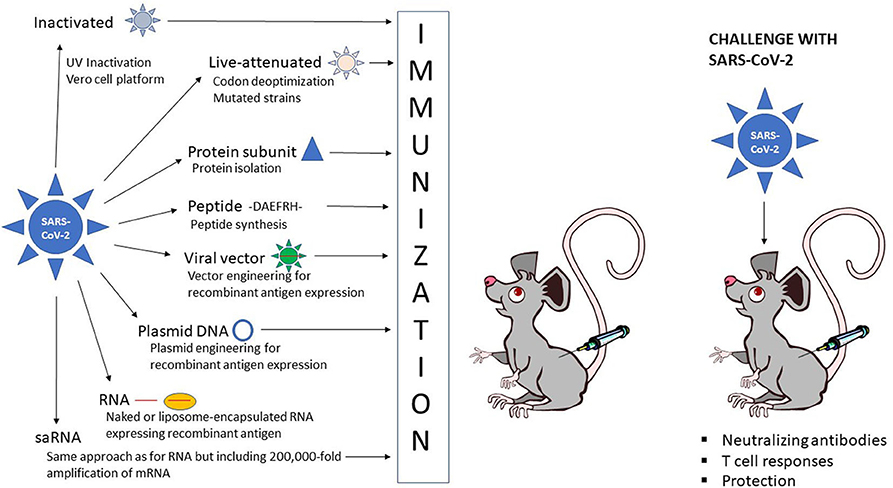
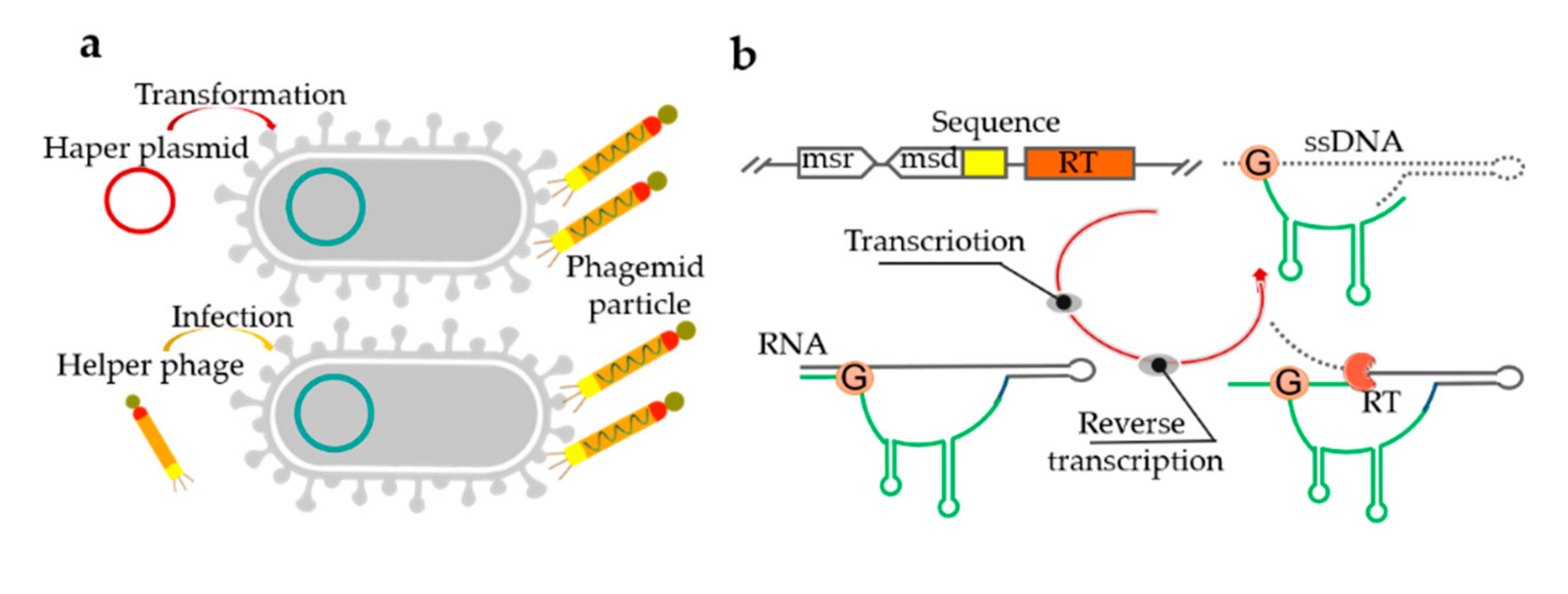
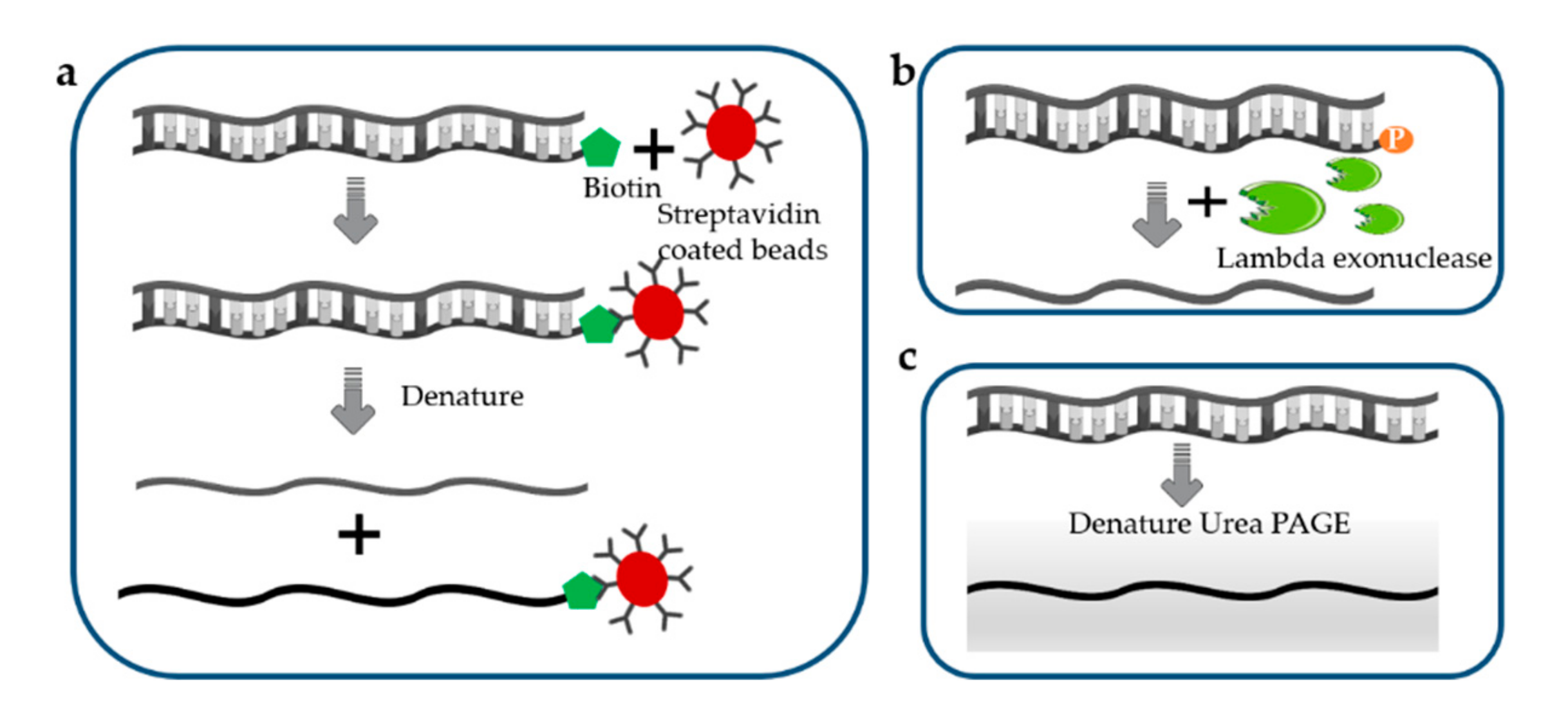
Potential application of synthetic biology in disease information recording and real-time monitoring

Genetic structure and dispersal in peripheral populations of a marine fish (Pacific cod, Gadus macrocephalus) and their importance for adaptation to climate change - Fisher - 2022 - Ecology and Evolution - Wiley Online Library

Figure 1 from A Novel msDNA (Multicopy Single-Stranded DNA) Strain Present in Yersinia frederiksenii ATCC 33641 Contig01029 Enteropathogenic Bacteria with the Genomic Analysis of It's Retron | Semantic Scholar

The Formation of the 2′,5′-Phosphodiester Linkage in the cDNA Priming Reaction by Bacterial Reverse Transcriptase in a Cell-free System - ScienceDirect

Structural basis for template switching by a group II intron-encoded non-LTR-retroelement reverse transcriptase | bioRxiv

The Formation of the 2′,5′-Phosphodiester Linkage in the cDNA Priming Reaction by Bacterial Reverse Transcriptase in a Cell-free System - ScienceDirect

Figure 1 from A Novel msDNA (Multicopy Single-Stranded DNA) Strain Present in Yersinia frederiksenii ATCC 33641 Contig01029 Enteropathogenic Bacteria with the Genomic Analysis of It's Retron | Semantic Scholar
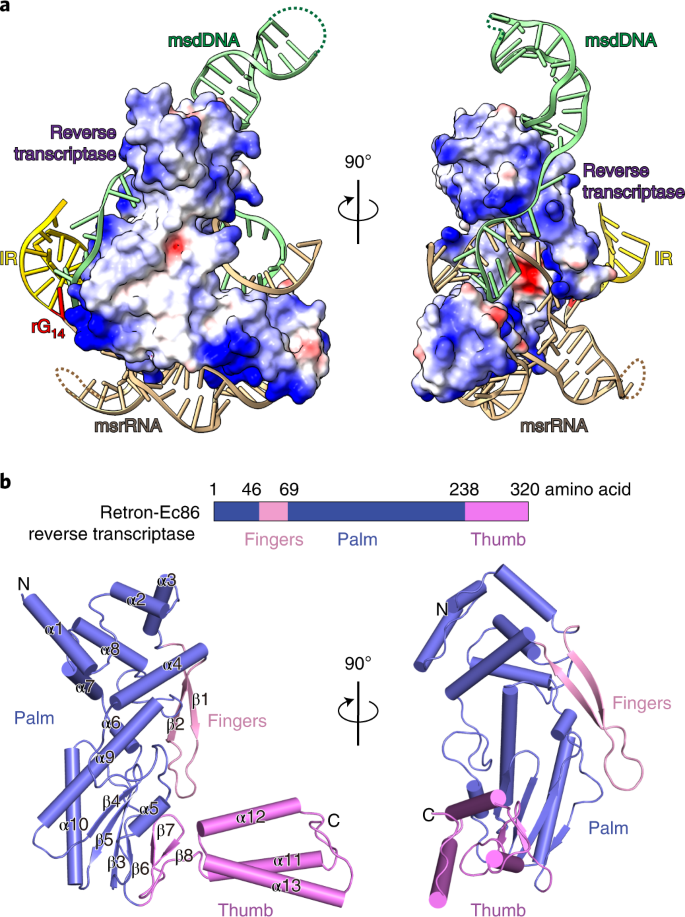
Cryo-EM structures of Escherichia coli Ec86 retron complexes reveal architecture and defence mechanism | Nature Microbiology

Structural basis for template switching by a group II intron-encoded non-LTR-retroelement reverse transcriptase | bioRxiv

The Formation of the 2′,5′-Phosphodiester Linkage in the cDNA Priming Reaction by Bacterial Reverse Transcriptase in a Cell-free System - ScienceDirect

Cryo-EM structures of Escherichia coli Ec86 retron complexes reveal architecture and defence mechanism | Nature Microbiology

The Formation of the 2′,5′-Phosphodiester Linkage in the cDNA Priming Reaction by Bacterial Reverse Transcriptase in a Cell-free System - ScienceDirect

Structural basis for template switching by a group II intron-encoded non-LTR-retroelement reverse transcriptase | bioRxiv