Vaccines | Free Full-Text | Estimating the Total Societal Cost of a Hexavalent Vaccine versus a Pentavalent Vaccine with Hepatitis B in South Korea

Recommendation for use of diphtheria and tetanus toxoids and acellular pertussis, inactivated poliovirus, <i xmlns="">Haemophilus influenzae</i> type b conjugate, and hepatitis B vaccine in infants
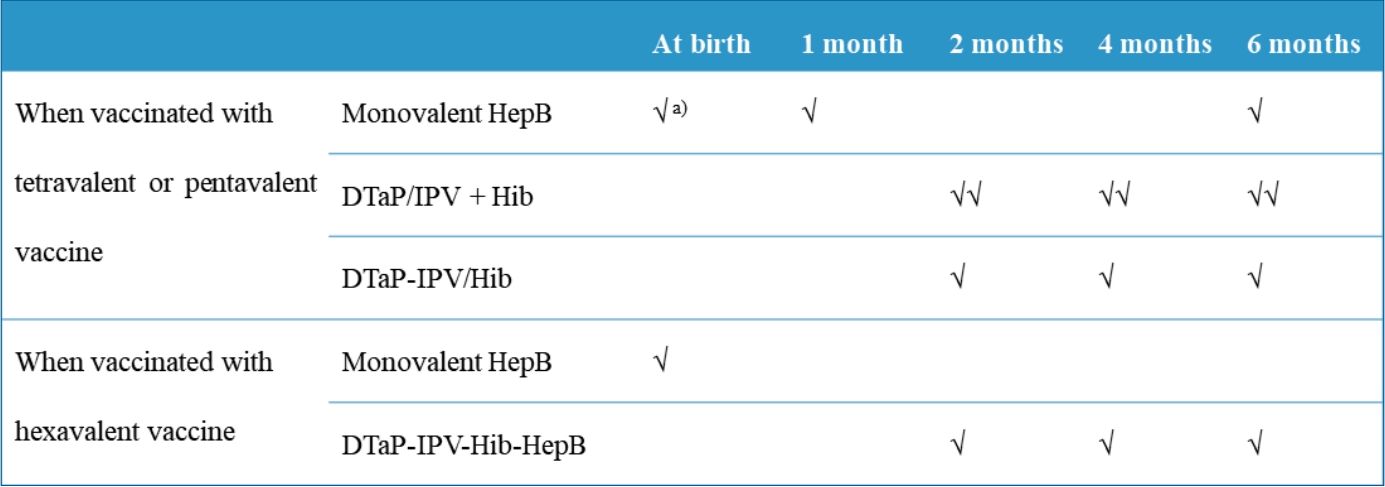
Recommendation for use of diphtheria and tetanus toxoids and acellular pertussis, inactivated poliovirus, Haemophilus influenzae type b conjugate, and hepatitis B vaccine in infants
![PDF] Immunogenicity and safety of an acellular pertussis, diphtheria, tetanus, inactivated poliovirus, Hib-conjugate combined vaccine (Pentaxim) and monovalent hepatitis B vaccine at 6, 10 and 14 weeks of age in infants in PDF] Immunogenicity and safety of an acellular pertussis, diphtheria, tetanus, inactivated poliovirus, Hib-conjugate combined vaccine (Pentaxim) and monovalent hepatitis B vaccine at 6, 10 and 14 weeks of age in infants in](https://d3i71xaburhd42.cloudfront.net/d3e50eb3c55e0b7aaef6c0fe1efdb53c3a90ac30/4-TableIII-1.png)
PDF] Immunogenicity and safety of an acellular pertussis, diphtheria, tetanus, inactivated poliovirus, Hib-conjugate combined vaccine (Pentaxim) and monovalent hepatitis B vaccine at 6, 10 and 14 weeks of age in infants in
Persistence of hepatitis B immune memory until 9–10 years of age following hepatitis B vaccination at birth and DTaP-IPV-HB-P
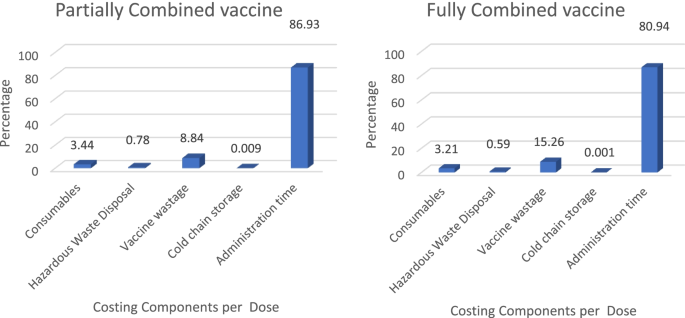
Economic impact of switching from partially combined vaccine “Pentaxim® and hepatitis B” to fully combined vaccine “Hexaxim®” in the Malaysian National Immunization Program | BMC Health Services Research | Full Text

PDF) Immunogenicity and safety of a pentavalent diphtheria, tetanus, acellular pertussis, inactivated poliovirus, Haemophilus influenzae type B conjugate combination vaccine (Pentaxim) with hepatitis B vaccine | Valsan Verghese - Academia.edu
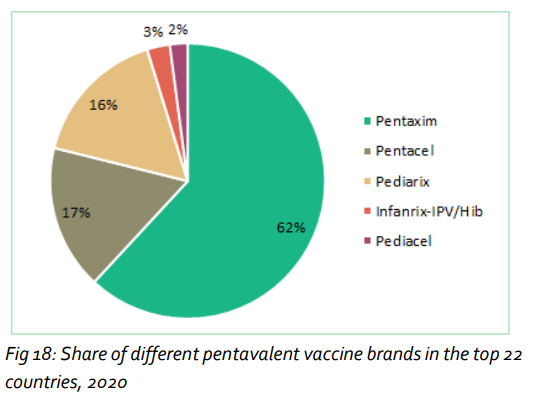
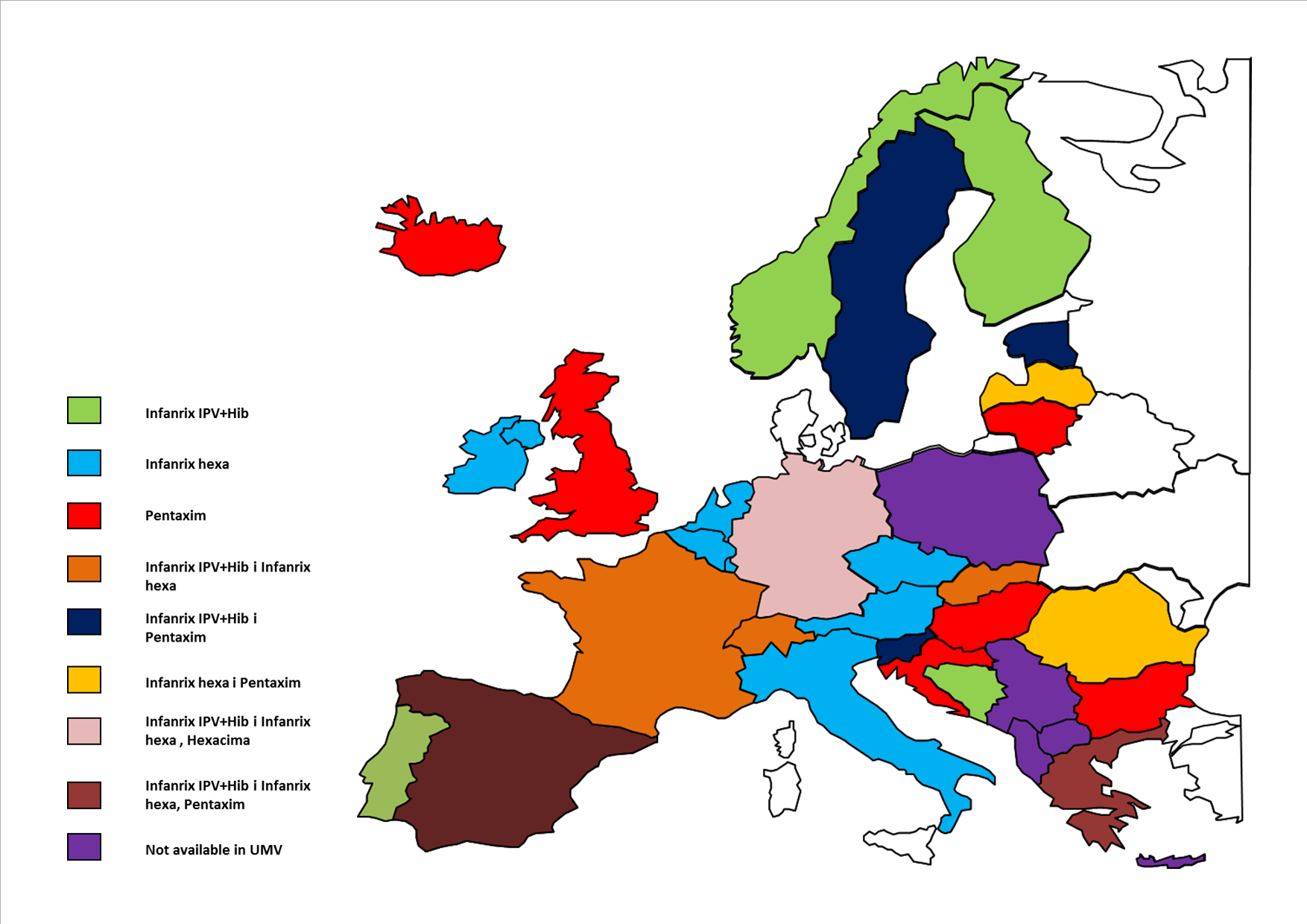
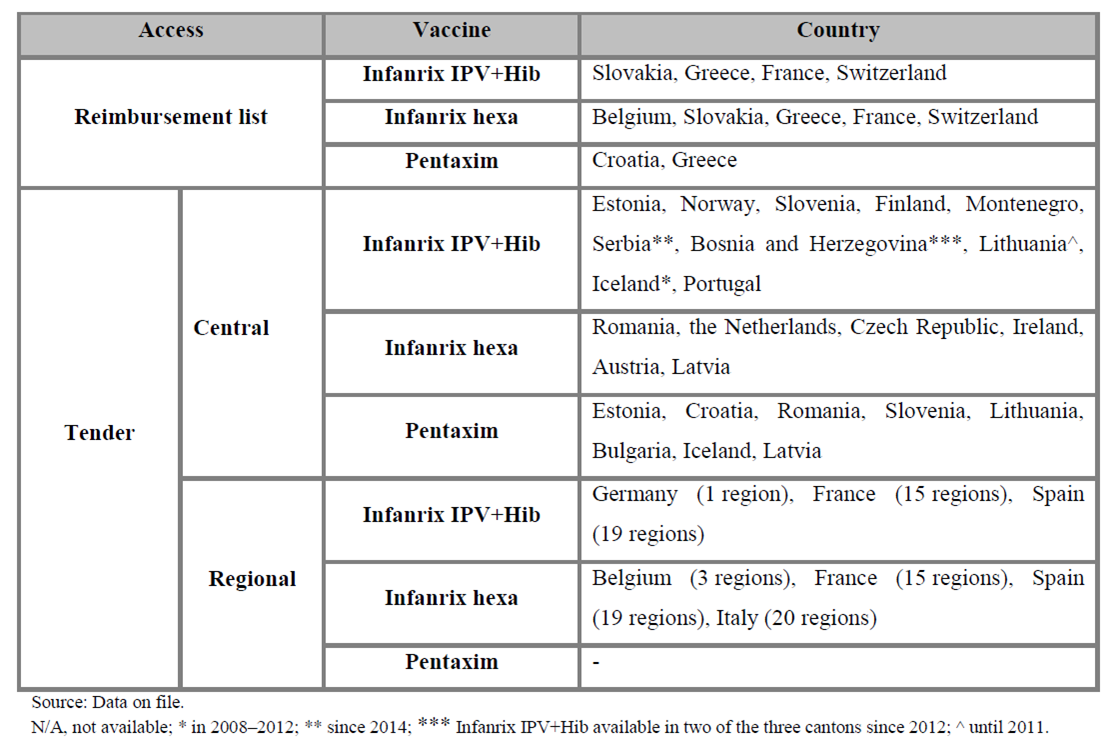
Sanofi Pasteur is the global market leader in all categories of aP-based pediatric combination vaccines